
FTC Sues to Stop Unlawful Blocking of Generic Androgel - IPWatchdog.com | Patents & Intellectual Property Law

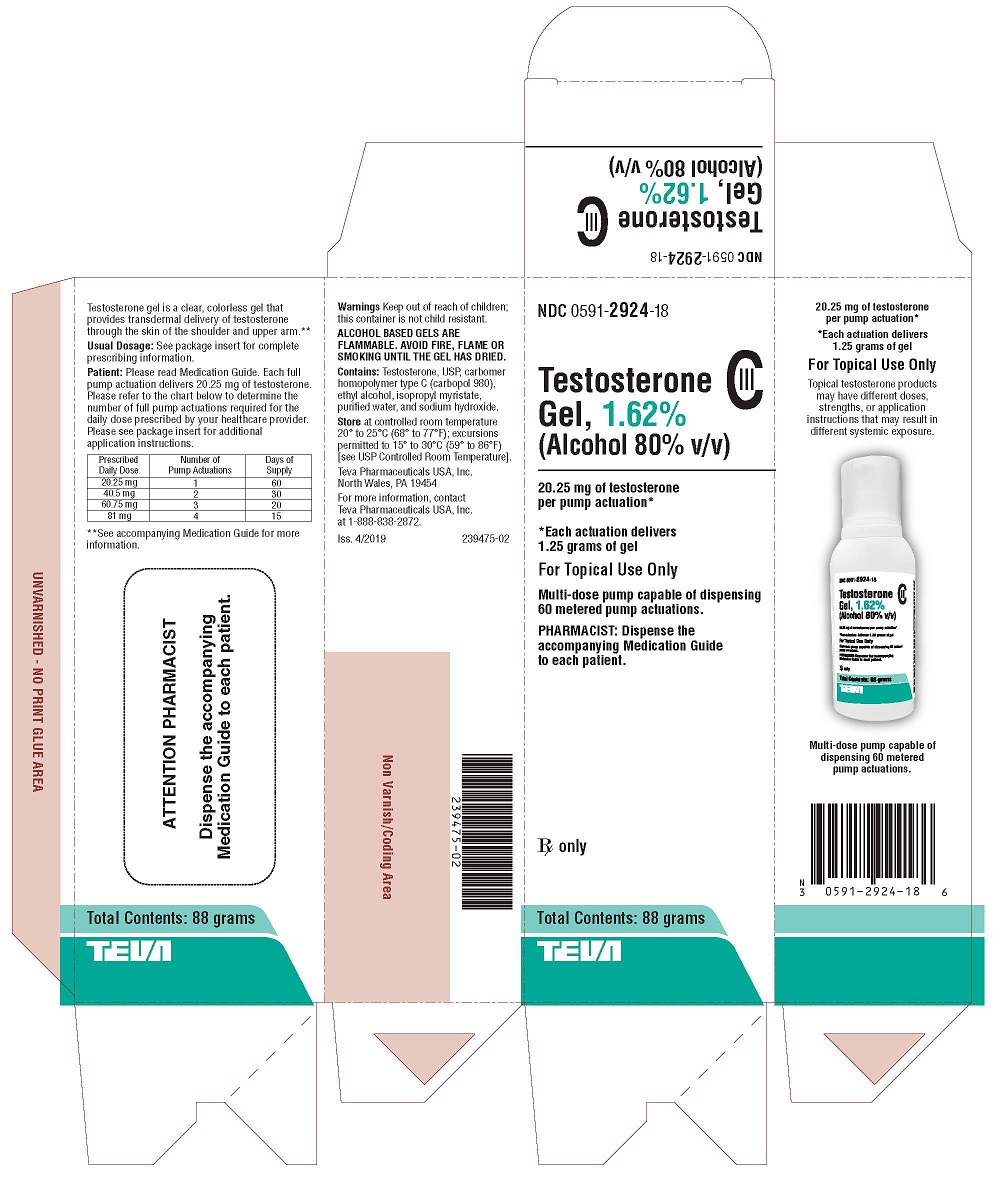
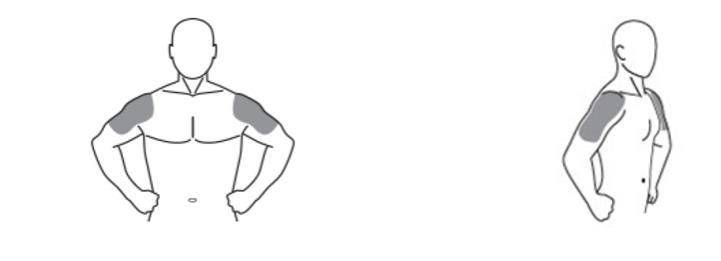
These highlights do not include all the information needed to use TESTOSTERONE GEL safely and effectively. See full prescribing information for TESTOSTERONE GEL . TESTOSTERONE gel 1.62% for topical use CIII Initial
Dissecting the importance of sex steroids balance for metabolic and reproductive health in men with Klinefelter syndrome: a rand







/arc-anglerfish-arc2-prod-pmn.s3.amazonaws.com/public/UB344NFNRNC2VHU73TRRKBOCDE.jpg)